Coronavirus drug from Japan to get first US trial in Massachusetts
Sanofi CEO: ‘Everybody gets access’ to potential coronavirus treatment if proven effective
Sanofi CEO Paul Hudson discusses the rapid progress being made in developing a coronavirus vaccine and moving the production of essential pharmaceutical supply from China to Europe.
Get all the latest news on coronavirus and more delivered daily to your inbox. Sign up here.
Continue Reading Below
Three major Massachusetts hospitals are testing a Japanese drug that could be used to treat novel coronavirus patients, The Boston Globe reported, citing a doctor involved in the trials.
The Food and Drug Administration approved a small, randomized trial of the antiviral drug called favipiravir at Massachusetts General Hospital and Brigham and Women's Hospital in Boston and UMass Memorial Medical Center in Worcester.
"We have to prioritize, and this absolutely rose to the top," Dr. Keith T. Flaherty, who is leading a team of experts at MGH to review COVID-19 clinical trials, told the Globe.
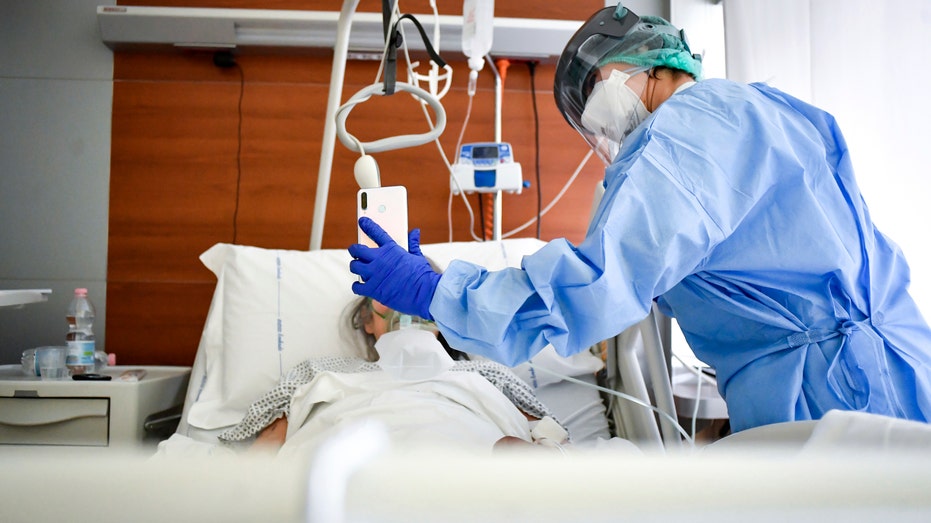
(AP Photo/Jacquelyn Martin, File) The Trump administration has reportedly been pushing the FDA to approve the drug known by its brand name Avigan and touted by Japanese Prime Minister Shinzo Abe as a possible treatment since late March, Politico reported, citing internal documents. TRUMP SAYS 'TOUGHEST' WEEKS AHEAD AS CORONAVIRUS SPREADS A Chinese official at the country's science and technology ministry said the drug was "clearly effective in treatment" and has a "high degree of safety" after the drug produced positive results in a trial involving 320 patients from Wuhan and Shenzhen, according to a March 16 report from The Guardian. GILEAD SCIENCES TO DONATE POSSIBLE CORONAVIRUS DRUG REMDESIVIR FOR EMERGENCY USE TESTING The Massachusetts trial will involve around 50 to 60 COVID-19 patients across the three hospitals. The patients will then be split into two groups — one of which will receive the normal standard of care associated with the drug while the other will receive the normal standard of care for COVID-19 patients, the Globe reported. A medical staffer holds up a phone in front of a COVID-19 patient for a video call with relatives at Bergamo’s Papa Giovanni XXIII hospital, northern Italy April 3. (Claudio Furlan/LaPresse via AP) Some researchers have concerns that the drug could have harmful side-effects, such as birth defects, officials told Politico. Dr. Robert W. Finberg, an infectious disease specialist at UMass Memorial, said that while the drug should not be prescribed to pregnant women, he told the Globe that the drug is "actually a very safe drug" and "looks promising." GET FOX BUSINESS ON THE GO BY CLICKING HERE Dr. Rochelle Walensky, MGH’s chief of infectious diseases, told the Globe that health care professionals haven't faced this kind of situation in which hospitals don't have "anything on the shelf to even try" for a disease treatment in "the last 20 years." "We are really motivated to understand how drugs might be helpful and work," she said. "It’s crushing to be on the front lines to see people getting worse in front of you and have nothing to offer." The president has been consistent in his optimism for a potential treatment drug called hydroxychloroquine, long used to treat malaria, rheumatoid arthritis and lupus, after very small preliminary studies suggested it might help prevent the coronavirus from entering cells and possibly help patients clear the virus sooner. GET FOX BUSINESS ON THE GO BY CLICKING HERE But the drug has major potential side effects, especially for the heart, and large studies are underway to see if it is safe and effective for treating COVID-19. CLICK HERE TO READ MORE ON FOX BUSINESS The United States is projected to surpass 400,000 confirmed COVID-19 cases Wednesday. Source: Read Full Article